Density of water at 20c4/15/2024 
Specific Weight (or force per unit volume) can be expressed asĪ g = acceleration of gravity (9.807, 32.174 under normal conditions) Specific Weight is defined as weight per unit volume. See also Thermophysical Properties of Air - density, viscosity, critical temperature and pressure, triple point, enthalpi and entropi, thermal conductivity and diffusicity. Molecular weights can be used to calculate Specific Gravity if the densities of the gas and the air are evaluated at the same pressure and temperature. Ρ air = density of air (normally at NTP - 1.204 ) The Specific Gravity can be calculated as Specific Gravity of gases is normally calculated with reference to air - and defined as the ratio of the density of the gas to the density of the air - at a specified temperature and pressure. Specific Gravity for some common Materials Specific Gravities common Substances Substance See also Thermophysical Properties of Water - Density, Freezing temperature, Boiling temperature, Latent heat of melting, Latent heat of evaporation, Critical temperature. Water is normally also used as reference when calculating the specific gravity for solids. SG of a fluid has the same numerical value as its density expressed in g/mL or Mg/m 3. Since Specific Gravity - SG - is dimensionless, it has the same value in the SI system and the imperial English system (BG). It is common to use the density of water at 4 oC (39 oF) as a reference since water at this point has its highest density of 1000 kg/m 3 or 1.940 slugs/ft 3. Ρ H2O = density of water - normally at temperature 4 oC Ρ substance = density of the fluid or substance Specific Gravity (Relative Density) - SG - is a dimensionless unit defined as the ratio of the density of a substance to the density of water - at a specified temperature and can be expressed as How to measure density of liquid petroleum products. 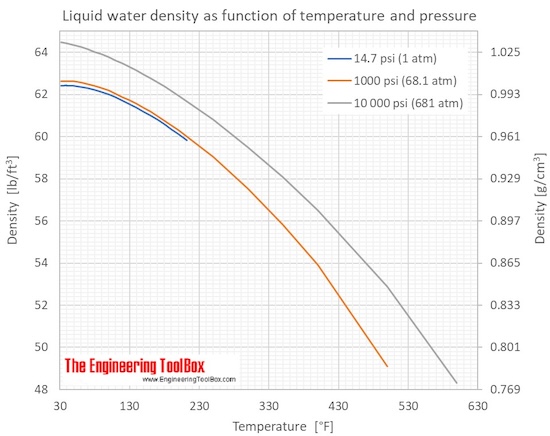
Air - Density, Specific Weight and Thermal Expantion Coefficient - variation with temperature and pressure, SI and Imperial units.Water - Density, Specific Weight and Thermal Expantion Coefficient - variation with temperature at 1, 68 and 680 atm, SI and Imperial units.Density is a physical property - constant at a given temperature and pressure - and may be helpful for identification of substances.īelow on this page: Specific gravity (relative density), Specific gravity for gases, Specific weight, Calculation examples On atomic level - particles are packed tighter inside a substance with higher density. See also Unit converter - mass and Unit converter - density Slugs can be multiplied with 32.2 for a rough value in pound-mass (lb m). Note that there is a difference between pound-force (lb f) and pound-mass (lb m). The Imperial (U.S.) units for density are slugs /ft 3 but pound-mass per cubic foot - lb m /ft 3 - is often used. What is weight and what is mass ? - the difference between weight and mass.Mass is a property and the SI unit for density is. Trending Questions Is a lever in a elevator? What are the names of the five platonic solids? What reading material is a common reason for resistance to receiving help is? What is the descriptive term does not apply to nuclear force? How many grams in a skein of wool? What is the crusing speed of a md87? Advantage and Disadvantage of Electromagnetic induction heat? How can you remember the difference between convex and concave lenses? What does a displaced meniscal tear mean? What is a way to say density? Why can gas move very fast and be compressed? What does 5 bar mean in watches? What are three ways you can change the magnetic field produced by a current? A simple machine requires this in order to operate? What is the order of magnitude for the thickness of a dollar bill? Why dont you see a beam of light from a flashlight refract when it travels across a room? What type of energy does a hand drill start with and what type of energy does it produce? Why do dryer sheets keep your clothes from sticking? Is it possible to see through clothes? A wavelength of 0.Density is defined as mass per unit volume.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
